Blood-Brain-Barrier (BBB) Integrity and Neurosafety Profiling with Primary Human OrganoReady® BBB HBMEC Model

Why This Is Important

Challenges

Need
MIMETAS’ Answer
.svg)
Organ Model
.svg)
Features
• Stable, functional and leak tight endothelial barrier
• Reproducible inflammatory response
• Robust monocyte adhesion assays
• Reliable toxicity read-outs for screening
.svg)
Offering
Custom CRO Services
Studying Vascular Inflammation
The human blood–brain barrier (BBB) plays a central role in maintaining brain homeostasis, yet inflammatory and drug-induced perturbations of BBB integrity present significant challenges in early CNS drug discovery and neurosafety assessment. Animal and in vitro models often lack human relevance or fail to capture key barrier functions, making early prediction of efficacy and toxicity difficult. Human-relevant models that provide robust readouts of barrier integrity, permeability, and inflammatory stress responses are needed to support faster, more reliable decision-making in drug development pipelines.
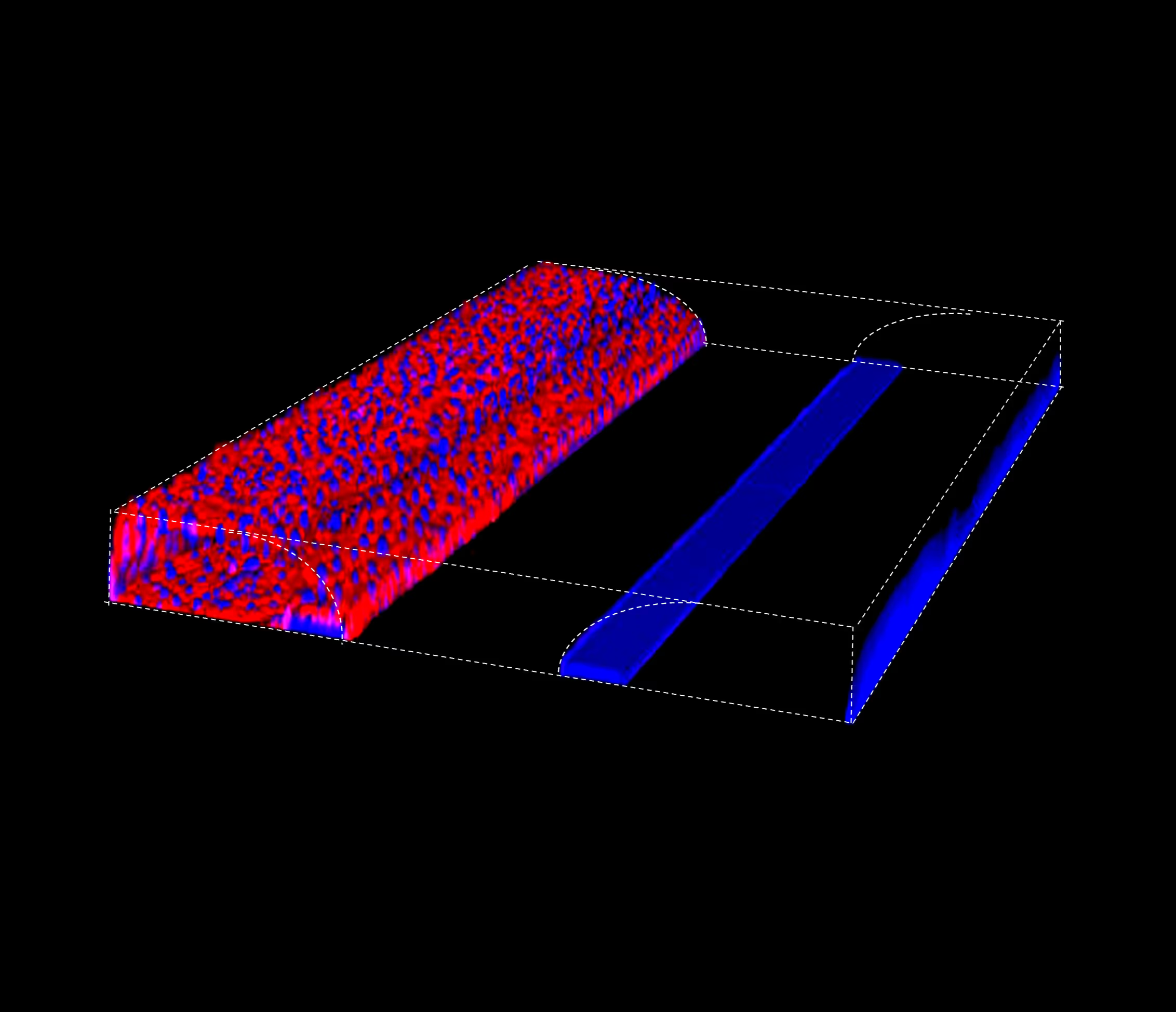
To address this need, we developed the OrganoReady BBB HBMEC. High-resolution imaging of the model reveals expresskey BBB markers, such as PECAM-1 and Claudin-5 (figure 1).
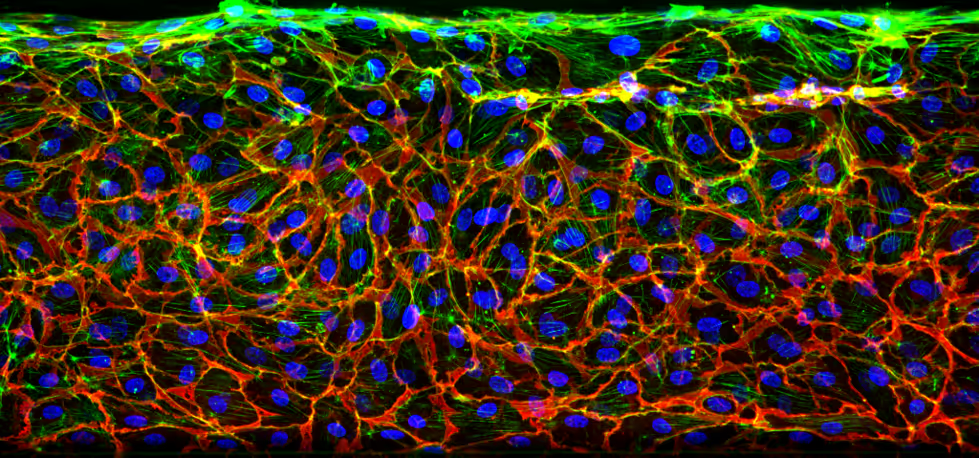
Neuroinflammation Modeling in an In Vitro BBB Model
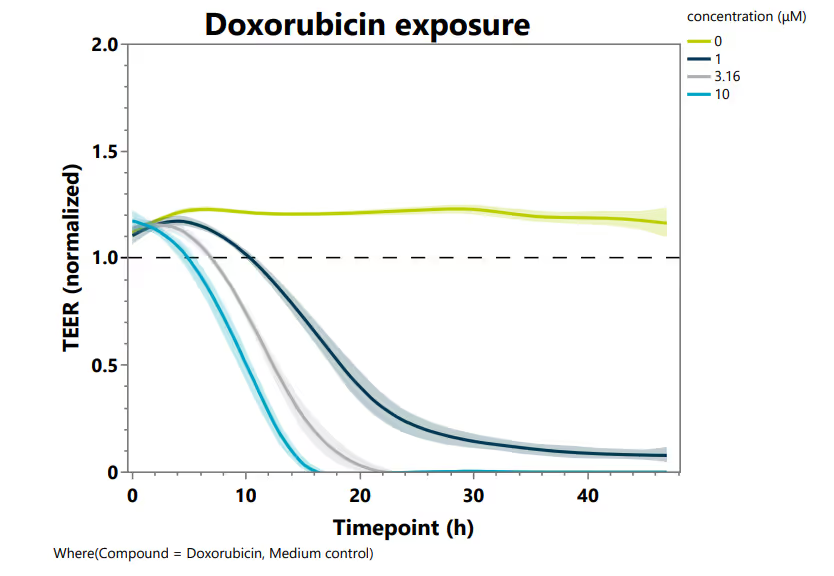
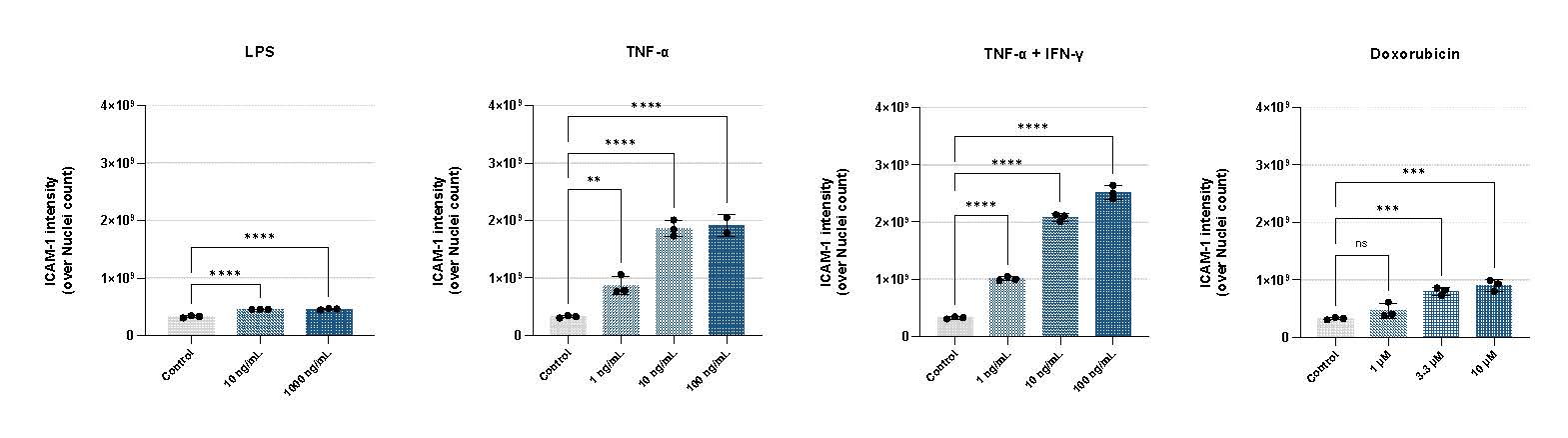
Cytokine exposure in our BBB model reproduces key features of neuroinflammation, enabling functional assessment of barrier integrity and analysis of molecular pathways underlying barrier disruption. The model shows a clear dose-dependent impairment of barrier function, with increasing cytokine concentrations leading to progressive decreases in TEER, measured using the OrganoTEER® device. The highest TNF-α and IL-1β cocktail reduced TEER by approximately 50% within 8 hours (Figure 2A). In parallel, cytokine stimulation resulted in reduced cell viability and decreased cell roundness, as quantified by VE-cadherin staining (Figure 3A, B). Furthermore, cytokine stimulation strongly upregulates ICAM-1 expression, demonstrating a robust inflammatory response (figure 4).

Assessing BBB Compound Safety
To assess compound-induced toxicity, we measured cell viability and cell roundness following exposure to increasing concentrations of staurosporine, a potent protein kinase inhibitor often used to induce endothelial stress. We observed a clear, concentration-dependent increase in LDH release, reaching a fourfold increase after 24 hours of treatment with 100 nM staurosporine (Figure 5A). Additionally, VE-cadherin–based analysis revealed a strong induction of cellular stress, reflected by decreased cell roundness (Figure 5B).
Summary
- Physiologically Relevant Vascular Inflammation: A perfusable 3D human blood vessel model with a stable, leak-tight endothelial barrier enables robust modeling of inflammatory activation, barrier disruption, and monocyte adhesion under flow.
- Predictive Safety and Toxicity Assessment: Real-time TEER measurements and dose-dependent endothelial responses provide sensitive, human-relevant readouts for vascular toxicity and drug-induced inflammation.
- Translational Immune–Endothelium Interactions: Controlled cytokine stimulation and PBMC perfusion support reproducible assessment of ICAM-1 upregulation and immune cell adhesion, bridging mechanistic insight with screening-ready throughput.
References
- Henein, M. Y., Vancheri, S.,Longo, G., & Vancheri, F. (2022). The role of inflammation incardiovascular disease. International journal of molecular sciences, 23(21),12906.
- Nam, U., Lee, S., Ahmad, A., Yi, H. G., &Jeon, J. S. (2024). Microphysiological systems as organ-specific in vitrovascular models for disease modeling. BioChip Journal, 18(3), 345-356.
Selected Resources
.avif)





