Gastrointestinal Safety Asessment with the Perfused OrganoReady® Colon Organoid Model


Why This Is Important

Challenges

Need
MIMETAS’ Answer
Screening Gastrointestinal Toxicity
Gastrointestinal (GI) toxicity can arise from multiple drug-induced mechanisms, including direct cellular toxicity, disruption of barrier function, and impaired mucus production. Drug mechanisms of action in the gut depend on interactions with different epithelial cell types, affecting barrier function and other key intestinal processes. The OrganoReady Colon Organoid tube contains several highly relevant gut cell types, including stem cells, correctly polarised enterocytes, and goblet cells (Figure 1).
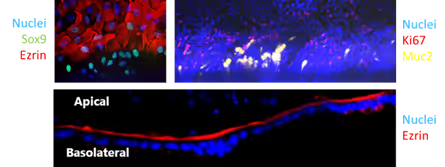
Figure 1. Immunofluorescent characterization demonstrating the presence of key intestinal epithelial cell types, including stem cells, enterocytes, and goblet cells, and correct apical-basolateral polarization within the 3D colon organoid tube.
Monitoring Barrier Integrity and Recovery in Real Time
Many drugs impair gut barrier integrity either directly, by disrupting cell–cell adhesion, or indirectly through cytotoxic effects. Real-time TEER measurements, obtained using the OrganoTEER® device, in the OrganoReady Colon Organoid enable robust, dose-dependent assessment of drug-induced barrier disruption as well as subsequent recovery (Figure 2).

Figure 1. Real-time TEER monitoring measurements showing concentration-dependent impairment of epithelial barrier integrity during treatment and subsequent recovery after compound withdrawal.
Functional Intestinal Read-outs
The colon organoid tubules recapitulate multiple essential epithelial functions. Upon exposure to vitamin D₃, gut cells show strong upregulation of CYP3A4, resulting in increased metabolic activity (Figure 3A). In addition, correct cellular polarisation and reporter expression enable highly reliable and physiologically relevant compound transport across the gut barrier (Figure 3B).

Figure 3. (A) Vitamin D₃-induced CYP3A4 activity measured via luciferin-based readout, with inhibition by ritonavir confirming metabolic specificity. (B) Time-dependent, directional compound transport across the epithelial barrier demonstrating polarized permeability and transporter-dependent modulation.
Predictive GI Safety Assessment
To evaluate compound-induced GI toxicity, the gut tubules were exposed to increasing compound concentrations for 5 days, followed by assessment of cell viability and barrier integrity (Figure 4A, B). This analysis was performed using a panel of 8 clinically GI-toxic drugs and 10 non-GI-toxic compounds. Using a margin of safety of 60 x Cmax, the model achieved a sensitivity of 75% and a specificity of 100%, demonstrating that the OrganoReady Colon is a powerful screening tool for GI safety assessment in drug development.

Figure 4. Concentration-response analysis of clinically characterized compounds demonstrating the model’s ability to distinguish GI-toxic from non-toxic drugs based on epithelial viability and barrier function metrics.
Summary
- Human-relevant intestinal complexity in a ready-to-use format: The OrganoReady Colon Organoid recapitulates key epithelial cell types, metabolic enzyme induction (CYP3A4), and physiologically relevant transport functions in a single platform.
- Predictive and robust GI safety performance: The model demonstrates 75% sensitivity and 100% specificity for clinically GI-toxic drugs, confirmed through real-time barrier integrity (TEER) and cell viability measurements.
- Actionable insights for safer drug development: By combining functional, metabolic, and barrier readouts, the system enables early, predictive screening of drug-induced GI toxicity to support informed safety assessment and decision-making.
References
- Federer, C., Yoo, M., & Tan, A. C. (2016). Big data mining and adverse event pattern analysis in clinical drug trials. Assay and Drug Development Technologies, 14(10), 557-566.
- Atkins, J. T., George, G. C., Hess, K., Marcelo-Lewis, K. L., Yuan, Y., Borthakur, G., ... & Hong, D. S. (2020). Pre-clinical animal models are poor predictors of human toxicities in phase 1 oncology clinical trials. British journal of cancer, 123(10), 1496-1501.
Selected Resources
.avif)

.svg)




