Modeling CAR T Cell Trafficking and Efficacy in Solid Tumors Using a Tumor–Vasculature-on-Chip

Why This Is Important

Challenges

Need
MIMETAS’ Answer
.svg)
Organ Model
.svg)
Features
• Live imaging of T-cell migration & killing
• Barrier-free T-cell movement
• Tunable ECM and cell composition
• Cytokine profiling & immunofluorescence readouts
.svg)
Offering
A Tumor-on-a-Chip Model
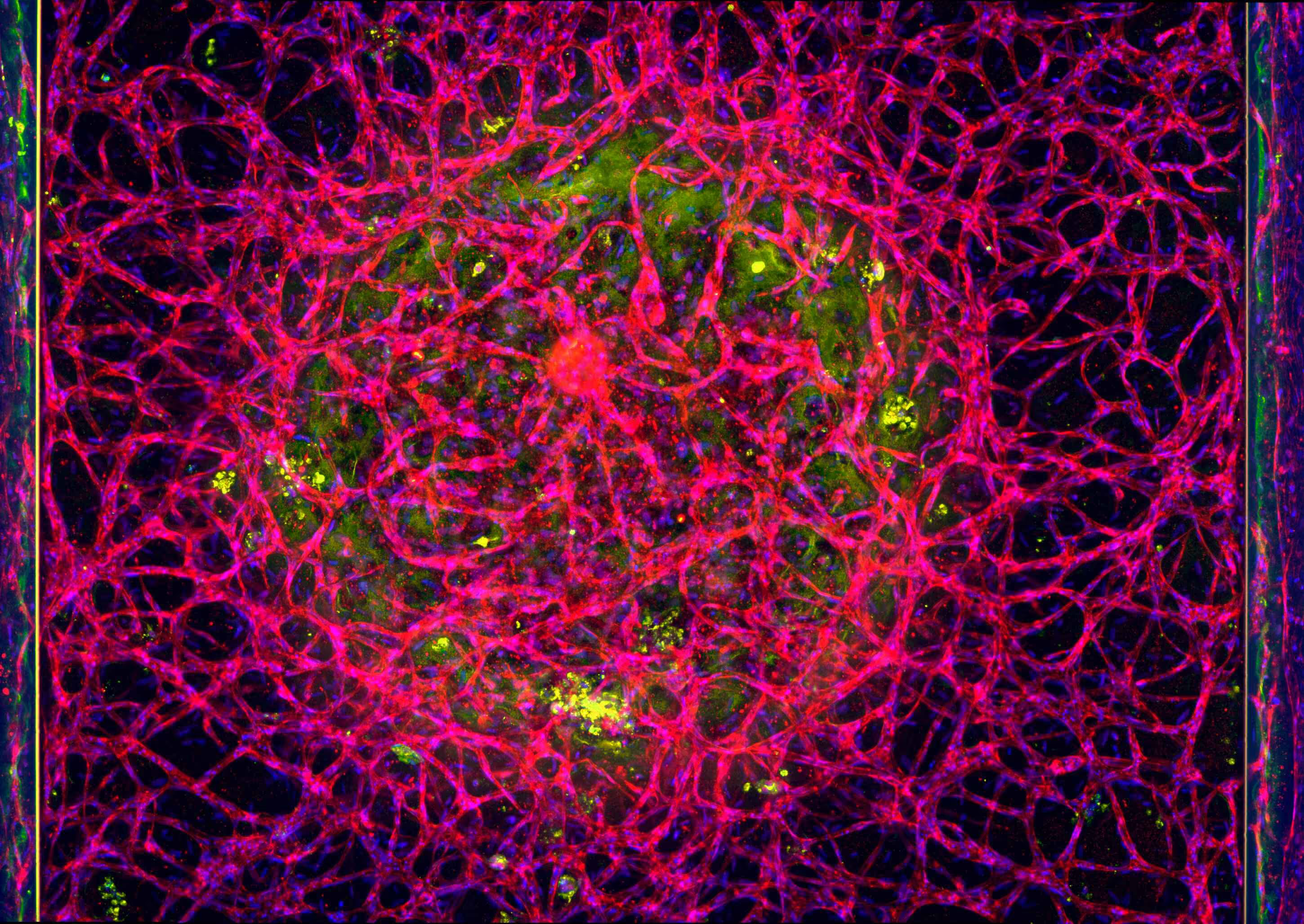
Solid tumors remain a major challenge for CAR T cell therapy due to limited infiltration, heterogeneous antigen expression, and therapy-induced toxicity. To study how CAR T cells reach and engage solid tumors, MIMETAS scientists developed an on-chip model to evaluate CAR T cell migration and killing capacity. ECM-embedded colorectal cancer (CRC) cells were incorporated into a model comprising endothelium and T cells. CAR T cells were perfused through the human endothelial vessel adjacent to ECM-embedded tumor cells (Figure 1).
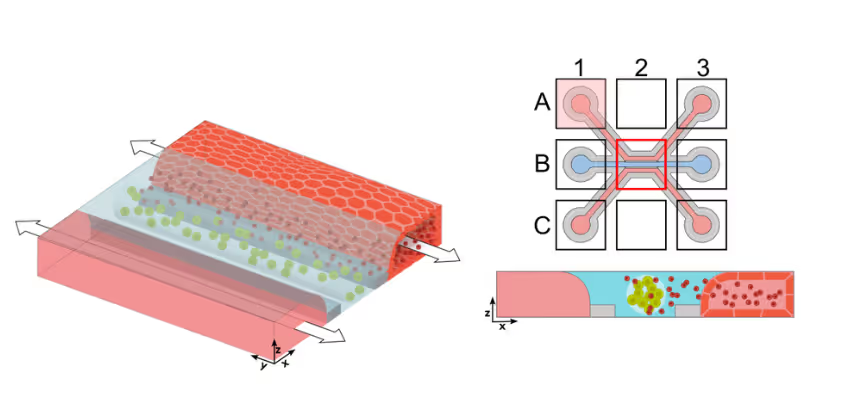
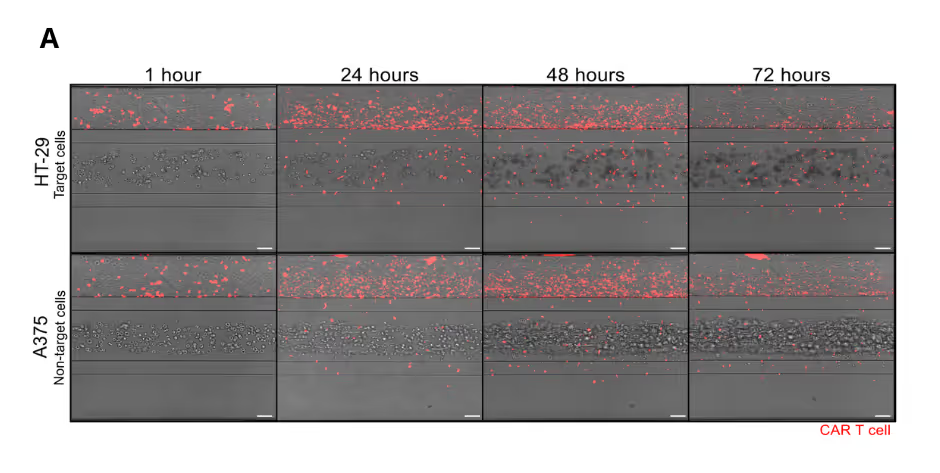
CAR T Cell Trafficking to Tumor Compartment
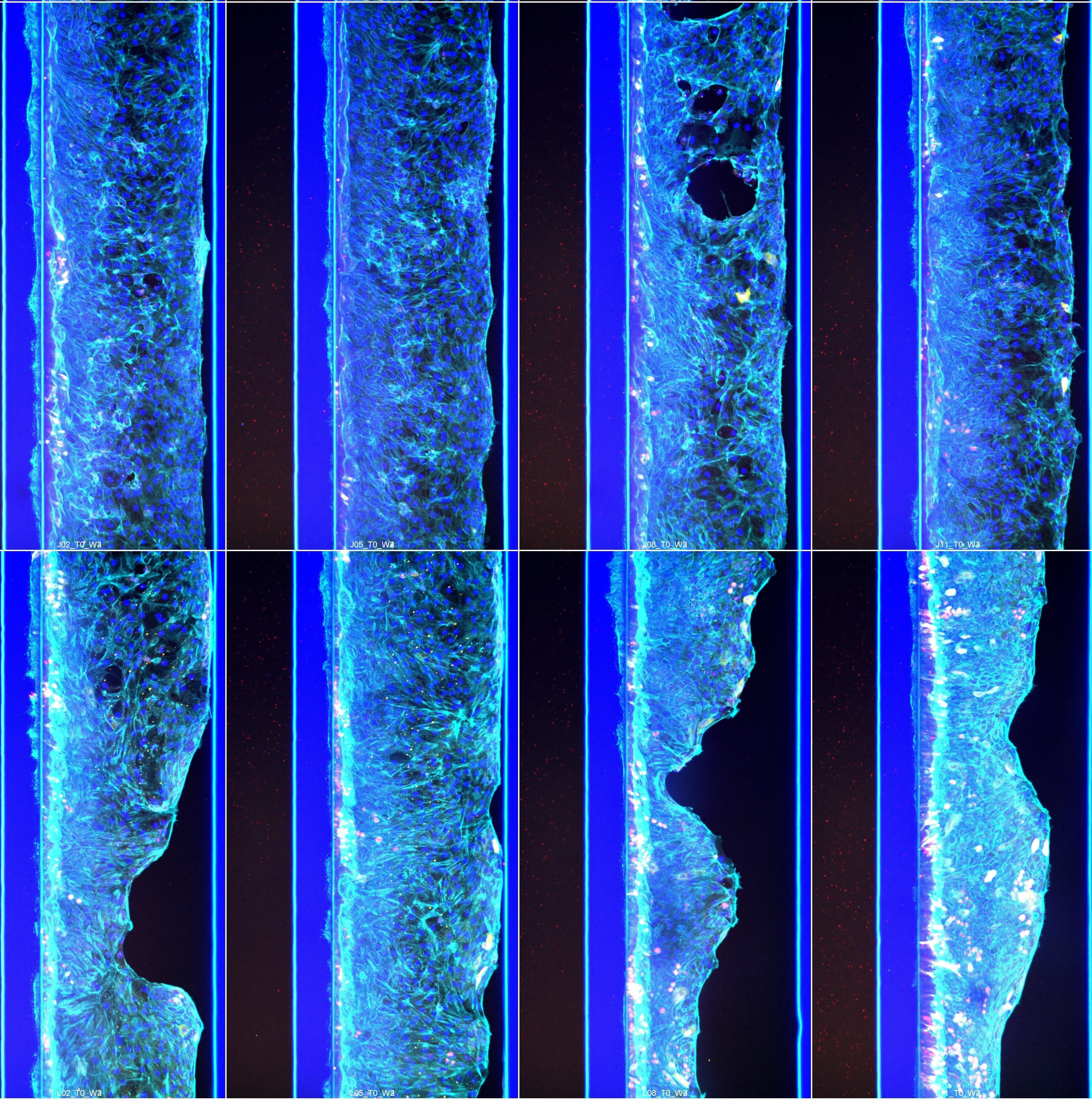
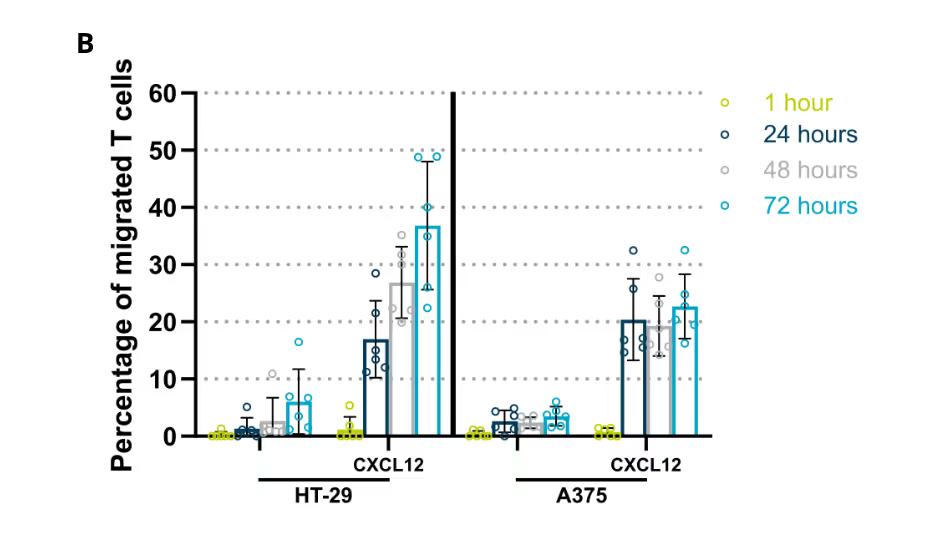
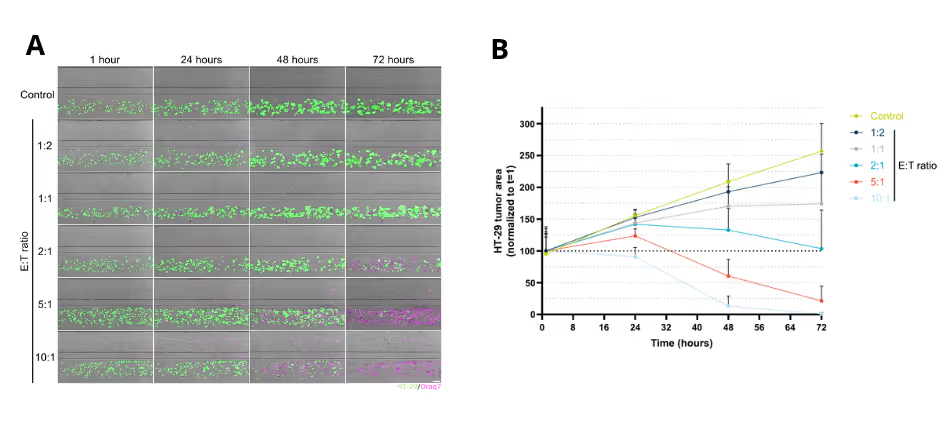
CAR T cells were perfused through a human endothelial vessel adjacent to ECM-embedded tumor cells, enabling assessment of vascular adhesion, transendothelial migration, and tumor infiltration. CAR T cell infiltration increased over time and was strongly enhanced by a CXCL12 chemokine gradient, indicating active and directed migration (Figure 2A). CAR T cells infiltrated both target and non-target tumor cell compartments. Quantitative analysis revealed distinct infiltration dynamics between these conditions, with differences in the fraction of CAR T cells entering the tumor compartment over time (Figure 2B). These results demonstrate that CAR T cell trafficking is influenced by tumor context and is not solely determined by target recognition.
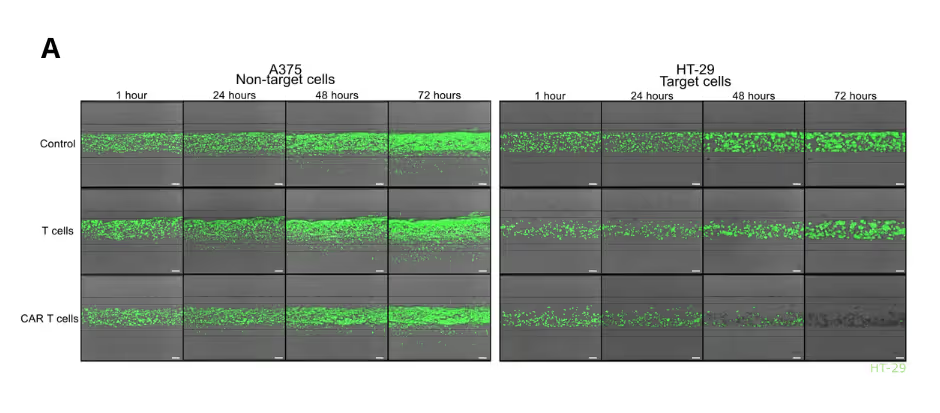
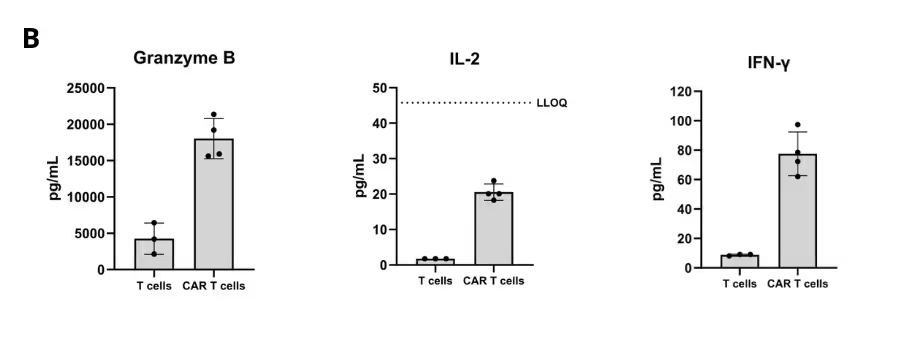
Solid Tumor Killing by CAR T Cells
The colon organoid tubules recapitulate multiple essential epithelial functions. Upon exposure to vitamin D₃, gut cells show strong upregulation of CYP3A4, resulting in increased metabolic activity (Figure 3A). In addition, correct cellular polarisation and reporter expression enable highly reliable and physiologically relevant compound transport across the gut barrier (Figure 3B).
Predictive GI Safety Assessment
To evaluate compound-induced GI toxicity, the gut tubules were exposed to increasing compound concentrations for 5 days, followed by assessment of cell viability and barrier integrity (Figure 4A, B). This analysis was performed using a panel of 8 clinically GI-toxic drugs and 10 non-GI-toxic compounds. Using a margin of safety of 60 x Cmax, the model achieved a sensitivity of 75% and a specificity of 100%, demonstrating that the OrganoReady Colon is a powerful screening tool for GI safety assessment in drug development.
Summary
- Simulation of Solid Tumor Microenvironment: Inclusion of factors such as immunosuppressive cells, soluble factors, and ECM stiffness.
- Accurate Immune Modeling: Real-time T-cell migration, engagement and cytokine release.
- Effective Construct Testing: Compare efficacy, and targeting precision of CAR T variants
References
- Chen Y, E C-Y, Gong Z-W, Liu S, Wang Z-X, Yang Y-S, et al. Chimeric antigen receptorengineered T-cell therapy for liver cancer. Hepatobiliary & Pancreatic Diseases International. 2018 Aug;17(4):301–9. doi:10.1016/j.hbpd.2018.05.005
- Siddiqui RS, Sardar M. A systematic review of the role of chimeric antigen receptor T (CAR-T) cell therapy in the treatment of solid tumors. Cureus. 2021 Apr 15; doi:10.7759/cureus.14494