Increased intestinal permeability has been described in a number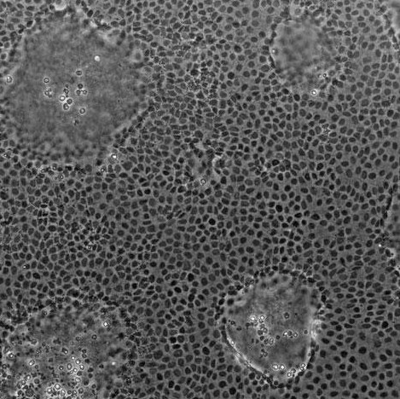 of diseases, including inflammatory bowel disorders (IBD)1, celiac disease2, and in conditions not primarily associated with the intestines.3 Even though the etiology of these diseases is far from clear, it is now widely accepted that the gut barrier plays an important role in health and disease.4 Studying intestinal permeability is critical to the development of therapeutics targeted at alleviating and avoiding the crippling symptoms associated with gut barrier disruption.
of diseases, including inflammatory bowel disorders (IBD)1, celiac disease2, and in conditions not primarily associated with the intestines.3 Even though the etiology of these diseases is far from clear, it is now widely accepted that the gut barrier plays an important role in health and disease.4 Studying intestinal permeability is critical to the development of therapeutics targeted at alleviating and avoiding the crippling symptoms associated with gut barrier disruption.
Mounting data from in vivo and in vitro studies show that intestinal permeability is influenced by stress, diet, microbial changes, and underlying genetic susceptibility. The complexity of intestinal permeability-related diseases makes it difficult to find suitable research models, let alone models that can be scaled up for high-throughput target validation and drug discovery purposes.5 While animal models have helped advance our understanding of IBD mechanisms, manipulating individual parameters such as epithelial barrier function and cell-type specific activation is challenging.5
When grown on permeable membranes, human colon adenocarcinoma (Caco-2) cells differentiate to form confluent monolayers, which display functional and structural characteristics of enterocytes (the cells that line the small and large intestine).6–8 Caco-2 cells can be used to study innumerable topics, including inflammatory triggers, bioavailability, permeability, and toxicity.9 Caco-2 cells are also considered to be the gold standard for in vitro prediction of intestinal drug permeability and oral drug absorption. However, creating intestinal permeability models with Caco-2 cells isn’t always as smooth-sailing as you might hope.
Here are seven problems you might be facing when creating your intestinal permeability model with Caco-2 cells:
1. Caco-2 cell tolerance of DMSO
Dimethylsulfoxide (DMSO) is a standard solvent used for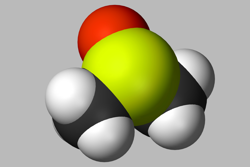 preparing stock solutions and is one of the principal additives in assay buffers.10 Although cytotoxic at high doses, it is often used in low concentrations as the control reagent during exposure, and in cryopreservation. The tolerance for DMSO varies across cells and transport proteins, and its possible effects on your results are worth bearing in mind; a recent epigenetic study revealed that low doses of DMSO induced major changes in DNA methylation in cardiac (but not hepatic) microtissues.11
preparing stock solutions and is one of the principal additives in assay buffers.10 Although cytotoxic at high doses, it is often used in low concentrations as the control reagent during exposure, and in cryopreservation. The tolerance for DMSO varies across cells and transport proteins, and its possible effects on your results are worth bearing in mind; a recent epigenetic study revealed that low doses of DMSO induced major changes in DNA methylation in cardiac (but not hepatic) microtissues.11
Above a certain threshold, DMSO can affect cell growth, protein stability and aggregation, and binding of drug compounds.10,12 Some protocols advise running a solvent tolerance test to ensure your results are not affected by the concentration of DMSO used in your cell culture experiment.6,10
2. Caco-2 cells taking too long to grow
If you’ve ever grizzled about your Caco-2 cells taking too long to grow, know that you are not alone. Slow-growing cells can be frustrating, especially when the problem significantly drags out your experiments, and limits the number of experiments you are able to complete.
too long to grow, know that you are not alone. Slow-growing cells can be frustrating, especially when the problem significantly drags out your experiments, and limits the number of experiments you are able to complete.
Troubleshooting can be time-consuming as there are many variables and possible reasons for slow growth; mycoplasma contamination and an overly dense feeder layer are two major possibilities.
3. Overcrowding of Caco-2 cells
The design of transport assays is based on the assumption that cells are organized in a monolayer, mimicking enterocytes in vivo. However, dome formation of Caco-2 cells can occur when a cell layer is pushed away from the culture dish by the accumulation of fluid beneath.13 This distortion can create fluid pockets, leading to uneven treatment distribution, and inadequate oxygen supply.14 These factors can compromise your transport assay results. Overcrowding can also occur when cells become too confluent, and differentiate before you are ready to run your assay. Subculturing cells before they have reached confluence can help cells to form a more homogenous and polarized monolayer.15 One protocol suggests subculturing cells when they have reached 50% of confluence, in contrast to an existing recommendation of 80%.15
4. Stability or instability over time
The number of times a cell culture is subcultured, or transferred from one vessel to another, is known as the “passage number”. Higher passage numbers and longer culture times can compromise genome stability and alter critical cell characteristics. Caco-2 cells are no exception; gene expression, phenotype and signaling pathways have been reported to drift over time in Caco-2 cells.16 Drifting gene expression levels can affect cell function and contribute to erroneous results.
Protocols generally recommend limiting continuous cell cultures to three months, or a particular number of passages. Excessive time in culture can also increase the risk of cell culture contamination, which is another reason to keep culture times on the shorter side where possible. There is no “magic number” of acceptable passages, and constant monitoring for changes in phenotype and cell density is essential.
5. Potential scalability for drug discovery
From a drug discovery perspective, models are needed that allow researchers to screen thousands of drug candidates. Conventional membrane insert-based systems are limited in their scalability on this level, and typically do not cater for 3D cellular models – an aspect that is desirable for improving physiological relevance. While you might have the ideal cell type for modeling intestinal permeability, having the potential to incorporate 3D models or other cell types would be highly beneficial.17
allow researchers to screen thousands of drug candidates. Conventional membrane insert-based systems are limited in their scalability on this level, and typically do not cater for 3D cellular models – an aspect that is desirable for improving physiological relevance. While you might have the ideal cell type for modeling intestinal permeability, having the potential to incorporate 3D models or other cell types would be highly beneficial.17
6. Research costs associated with compounds, media, and reagents
Whenever you are troubleshooting issues with culturing your Caco-2 cells, you could potentially be wasting huge amounts of money on expensive compounds – not to mention copious amounts of fetal bovine serum and other reagents. Any technology that reduces your reagent, cell, and time consumption can significantly reduce research and development costs.
7. The need for specialized instruments and equipment
Modern drug discovery approaches rely on high-resolution kinetic measurements and image-based readouts. However, conventional transwell models are poorly suited to the instruments and imagers required for kinetic measurements.17 Having a platform compatible with automated plate readers, microscopes, and other technologies could help researchers to extract critical information about intestinal permeability from their Caco-2 cells.
Some research groups working with Caco-2 cells might find they have a shortage of laboratory personnel with the specialized skills required for setting up desired experiments. Certain research models may require staff to be knowledgeable in microfluidics, or experienced in assessing barrier integrity. The transepithelial electrical resistance (TEER) is a measure of the integrity of a monolayer corresponding to barrier function, with differentiated polarized cells that have formed tight junctions.18 TEER is used to gauge the effect of different exposure times and concentrations. Without prior experience, navigating the literature surrounding TEER techniques19 can be challenging.
One Nature Protocols paper states that “Given the necessary care and attention, most cell lines are easy to maintain and grow”.20 Despite this, most researchers still find there are many obstacles to navigate when working with Caco-2 cells. If Caco-2 cells are critical to your work, be sure to check this blog post where we will show you how you can grow your intestinal permeability models in less than five days.
Learn more about MIMETAS organ-on-a-chip technology >>
Related resourcesKeep me informed
References
- Vivinus-Nébot, M., Frin-Mathy, G., Bzioueche, H., Dainese, R., Bernard, G., Anty, R., Filippi, J., Saint-Paul, M. C., Tulic, M. K., Verhasselt, V., Hébuterne, X., & Piche, T. (2014). Functional bowel symptoms in quiescent inflammatory bowel diseases: Role of epithelial barrier disruption and low-grade inflammation. Gut, 63(5), 744–752.
- Heyman, M., Abed, J., Lebreton, C., & Cerf-Bensussan, N. (2012). Intestinal permeability in coeliac disease: Insight into mechanisms and relevance to pathogenesis. Gut, 61(9), 1355–1364.
- González-González, M., Díaz-Zepeda, C., Eyzaguirre-Velásquez, J., González-Arancibia, C., Bravo, J. A., & Julio-Pieper, M. (2019). Investigating Gut Permeability in Animal Models of Disease. Frontiers in Physiology, 9, 1962.
- Vancamelbeke, M., & Vermeire, S. (2017). The intestinal barrier: A fundamental role in health and disease. Expert Review of Gastroenterology & Hepatology, 11(9), 821–834.
- Beaurivage, C., Naumovska, E., Chang, Y., Elstak, E., Nicolas, A., Wouters, H., van Moolenbroek, G., Lanz, H., Trietsch, S., Joore, J., Vulto, P., Janssen, R., Erdmann, K., Stallen, J., & Kurek, D. (2019). Development of a Gut-on-a-Chip Model for HighThroughput Disease Modeling and Drug Discovery. International Journal of Molecular Sciences, 20(22), 5661.
- Hubatsch, I., Ragnarsson, E. G. E., & Artursson, P. (2007). Determination of drug permeability and prediction of drug absorption in Caco-2 monolayers. Nature Protocols, 2(9), 2111–2119.
- Hilgers, A., Conradi, R., & Burton, P. (1990). Caco-2 cell monolayers as a model for drug transport across the intestinal mucosa. Pharmaceutical Research, 7(9), 902–910.
- Jumarie, C., & Malo, C. (1991). Caco-2 cells cultured in serum-free medium as a model for the study of enterocytic differentiation in vitro. Journal of Cellular Physiology, 149(1), 24–33.
- Maxnah, I. Jr. (1999). The use of Caco-2 cells as an in vitro method to study bioavailability of iron. Malaysian Journal of Nutrition, 5(1), 31–45.
- Tjernberg, A., Markova, N., Griffiths, W. J., & Hallén, D. (2006). DMSO-Related Effects in Protein Characterization. Journal of Biomolecular Screening, 11(2), 131–137.
- Verheijen, M., Lienhard, M., Schrooders, Y., Clayton, O., Nudischer, R., Boerno, S., Timmermann, B., Selevsek, N., Schlapbach, R., Gmuender, H., Gotta, S., Geraedts, J., Herwig, R., Kleinjans, J., & Caiment, F. (2019). DMSO induces drastic changes in human cellular processes and epigenetic landscape in vitro. Scientific Reports, 9(1), 4641.
- Rodríguez-Burford, C., Oelschlager, D. K., Talley, L. I., Barnes, M. N., Partridge, E. E., & Grizzle, W. E. (2003). The use of dimethylsulfoxide as a vehicle in cell culture experiments using ovarian carcinoma cell lines. Biotechnic & Histochemistry, 78(1), 17–21.
- Misfeldt, D., Hamamoto, S., & Pitelka, D. (1976). Transepithelial transport in cell culture. Proceedings of the National Academy of Sciences, 74(3), 1212–1216.
- von Köckritz-Blickwede, M., Zeitouni, N., Fandrey, J., & Naim, H. Y. (2015). Measuring oxygen levels in Caco-2 cultures. Hypoxia, 53.
- Natoli, M., Leoni, B. D., D’Agnano, I., D’Onofrio, M., Brandi, R., Arisi, I., Zucco, F., & Felsani, A. (2011). Cell growing density affects the structural and functional properties of Caco-2 differentiated monolayer. Journal of Cellular Physiology, 226(6), 1531–1543.
- Briske-Anderson, M. J., Finley, J. W., & Newman, S. M. (1997). The Influence of Culture Time and Passage Number on the Morphological and Physiological Development of Caco-2 Cells. Experimental Biology and Medicine, 214(3), 248–257.
- Trietsch, S. J., Naumovska, E., Kurek, D., Setyawati, M. C., Vormann, M. K., Wilschut, K. J., Lanz, H. L., Nicolas, A., Ng, C. P., Joore, J., Kustermann, S., Roth, A., Hankemeier, T., Moisan, A., & Vulto, P. (2017). Membrane-free culture and real-time barrier integrity assessment of perfused intestinal epithelium tubes. Nature Communications, 8(1), 262.
- Sakharov, D., Maltseva, D., Knyazev, E., Nikulin, S., Poloznikov, A., Shilin, S., Baranova, A., Tsypina, I., & Tonevitsky, A. (2019). Towards embedding Caco-2 model of gut interface in a microfluidic device to enable multi-organ models for systems biology. BMC Systems Biology, 13(S1), 19.
- Srinivasan, B., Kolli, A. R., Esch, M. B., Abaci, H. E., Shuler, M. L., & Hickman, J. J. (2015). TEER Measurement Techniques for In Vitro Barrier Model Systems. Journal of Laboratory Automation, 20(2), 107–126.
- Masters, J. R., & Stacey, G. N. (2007). Changing medium and passaging cell lines. Nature Protocols, 2(9), 2276–2284
Blog post written by Michele Wilson at choicesciencewriting.com
Learn more
- [Application note] Comparison of the OrganoPlate® and the Transwell® platform for in vitro intestinal permeability assays
- [Protocol] Caco-2 Seeding in OrganoPlate® 3-lane
- [Blog Post] How To Grow Intestinal Permeability Models in 4 Days
- [Infographic] Why You Can Stop Using Plate Inserts For Intestinal Permeability Models
- [Infographic] Methods to Assess Barrier Integrity and Permeability in 3D tissue models
- [Webinar] Caco-2 Modeling
- [Webinar] OrganoTEER®: Know Your Barriers. Fast
- [Product] OrganoTEER®
